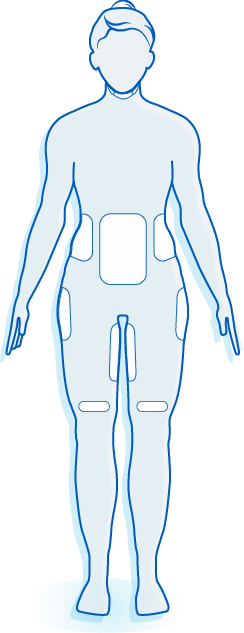
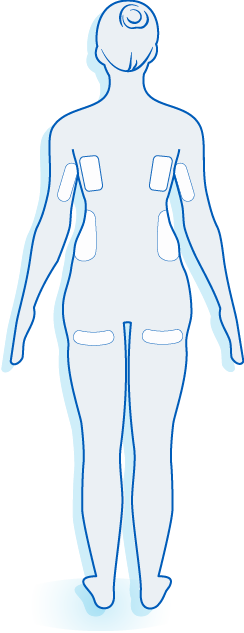
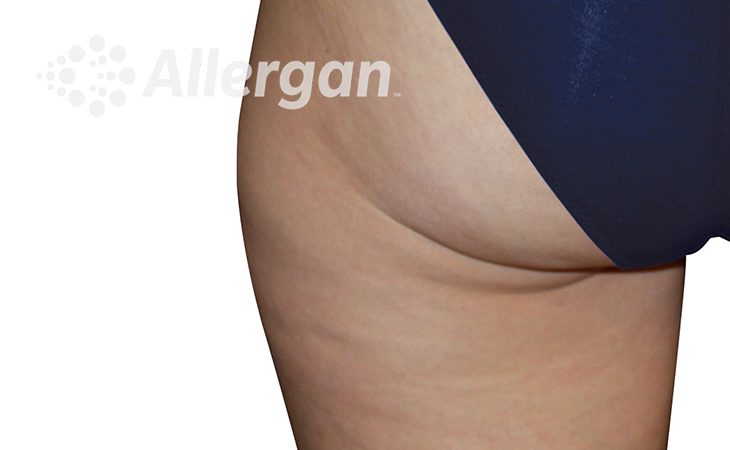
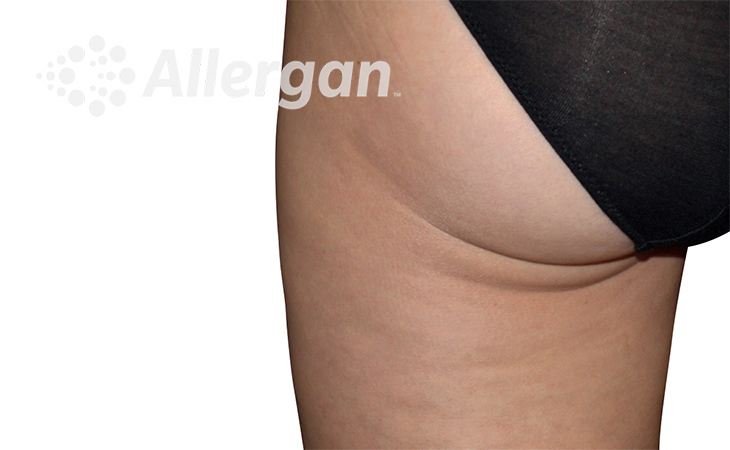
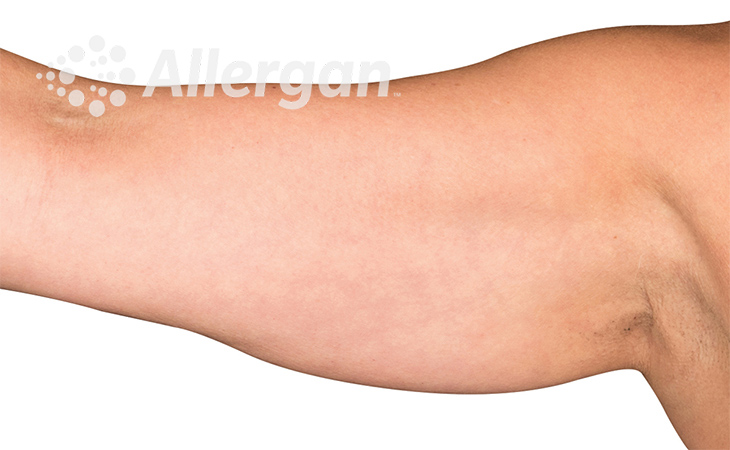
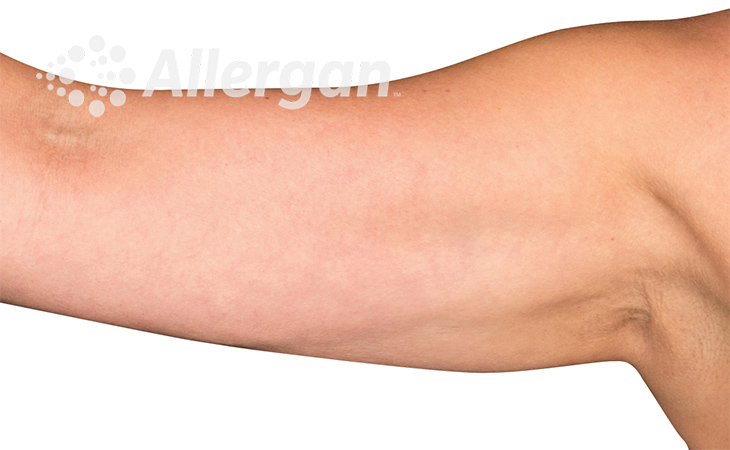
The World’s Original Fat Reduction Technology
“Cryolipolysis”1
an Advanced Fat Freezing Technology that freezes body fat
cells away
Over 11 million
treatment done worldwide2
Impressive scientific evidence
+70 scientific publications supported5
Non-invasive,
Lasting results,
Minimal downtime11, 12
The new CoolSculpting® Elite makes every
Details counts for your
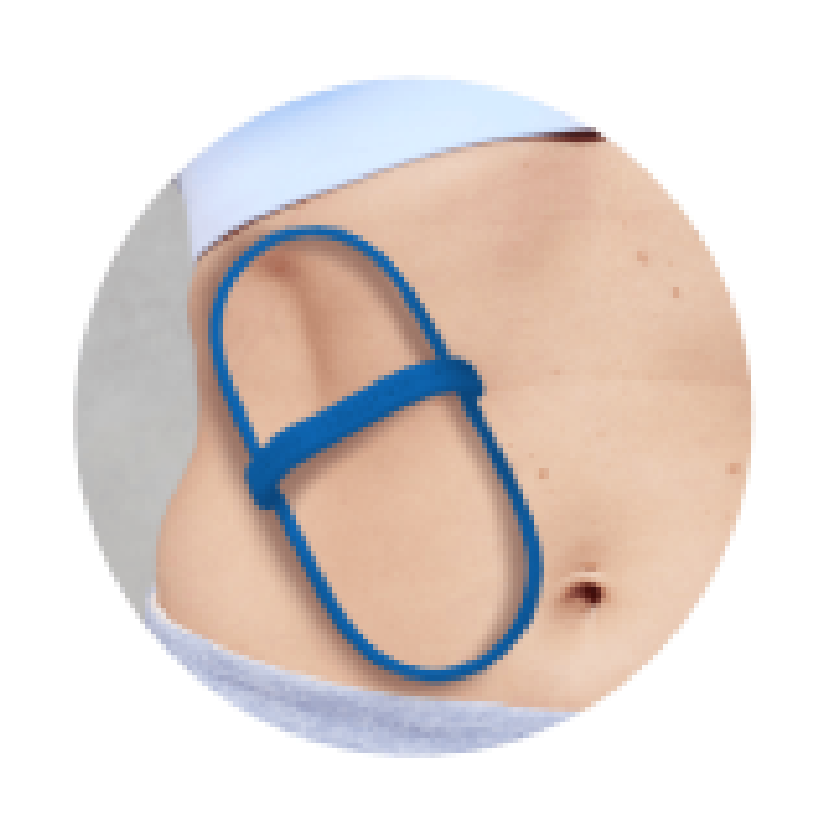
Non-invasive Fat Reduction Treatment
EVERY Hour COUNTS
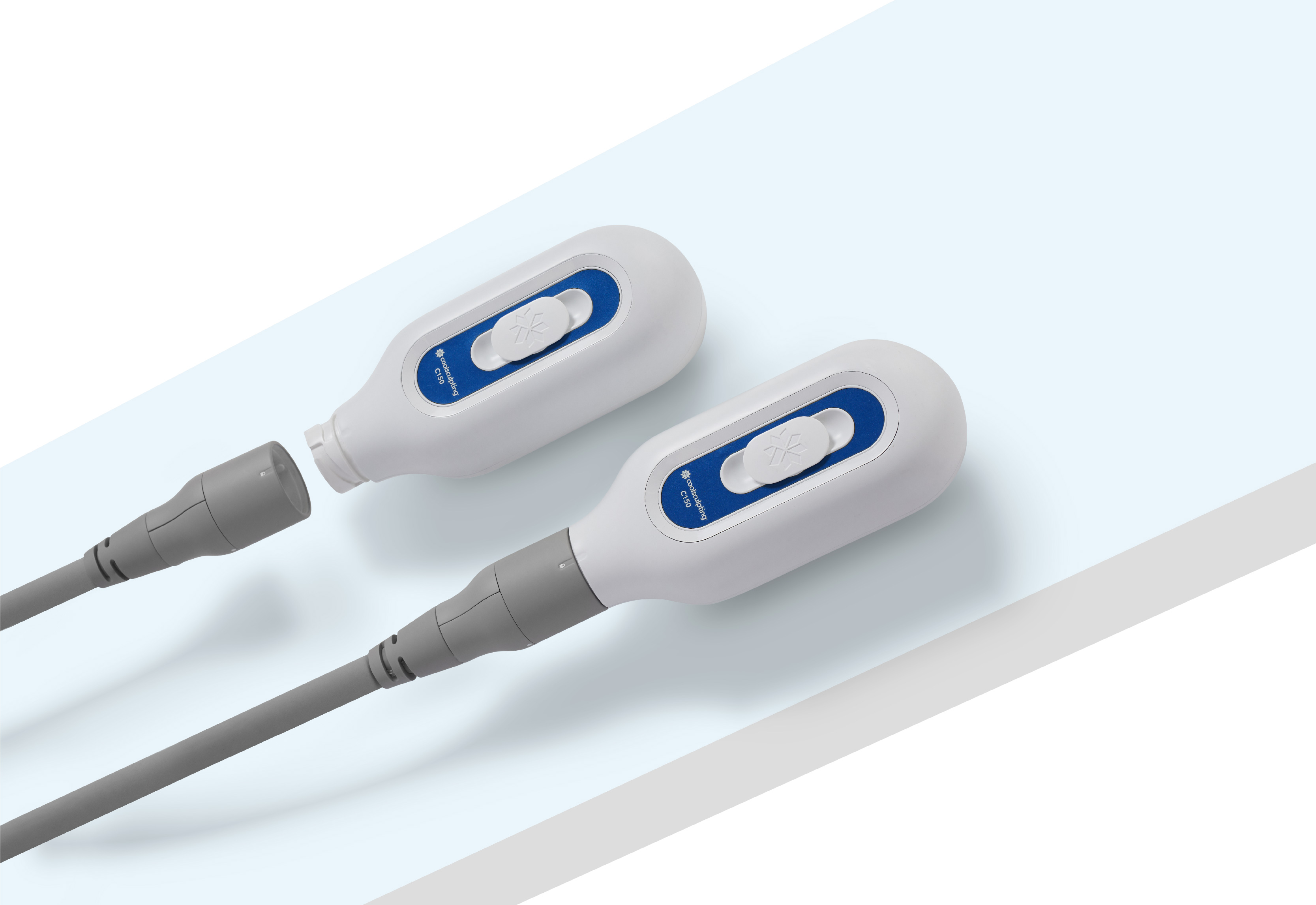
POWERED FOR
DUALITY
New CoolSculpting® Elite gives you beyond the results, but also efficiency.



1 machine x
2 applicators
As fast as 35mins
in one treatment session9.
Receive twice the result in single treatment session9.
EVERY Outcome COUNTS

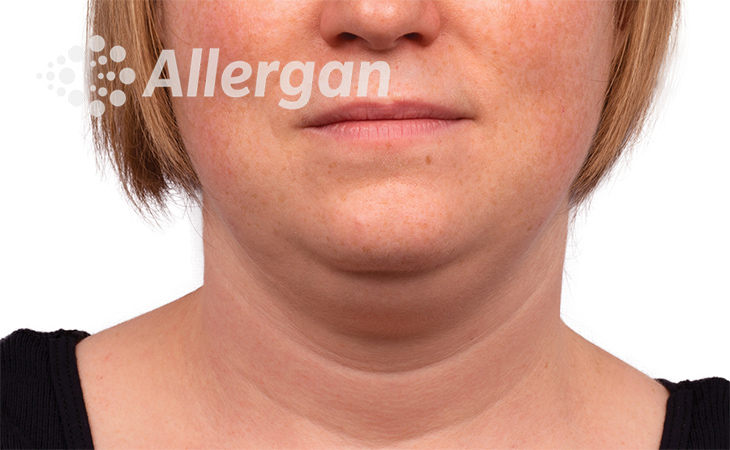
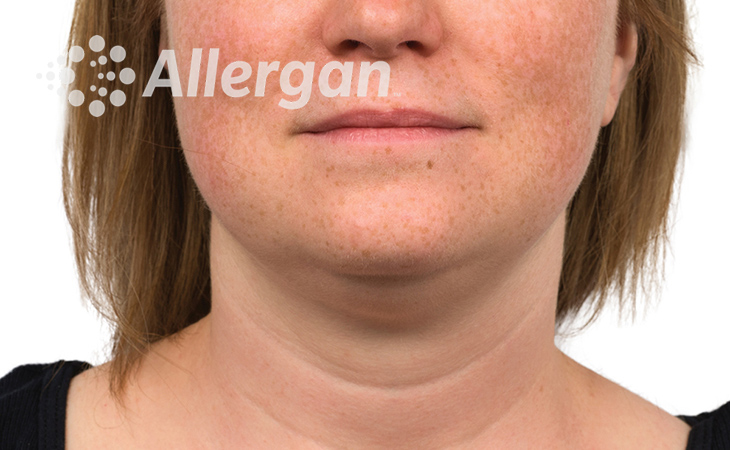
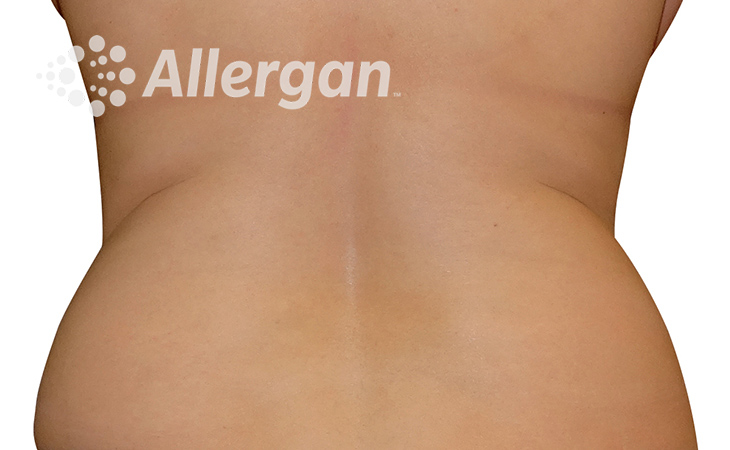
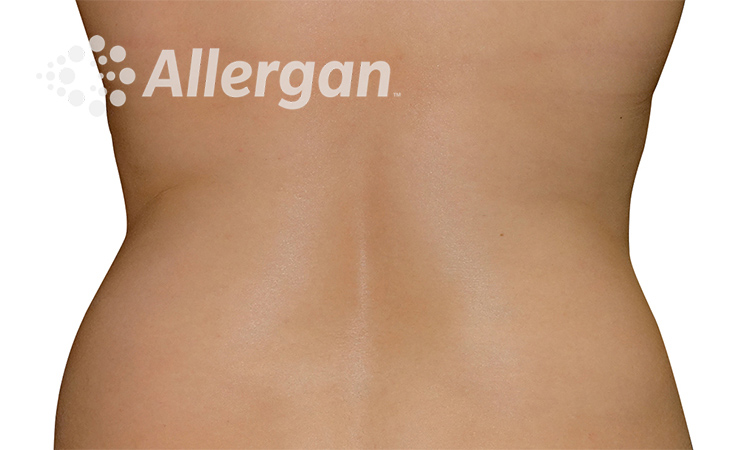
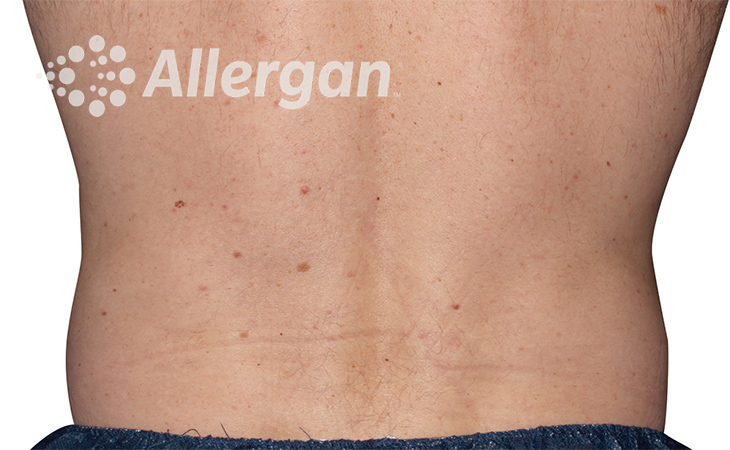
Fat reduction
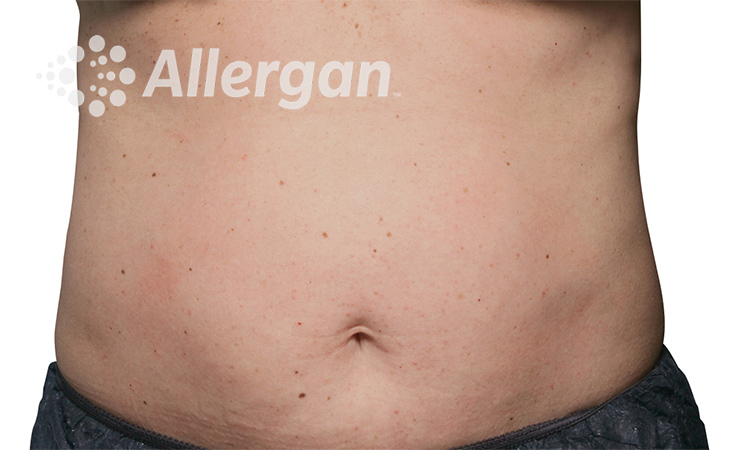
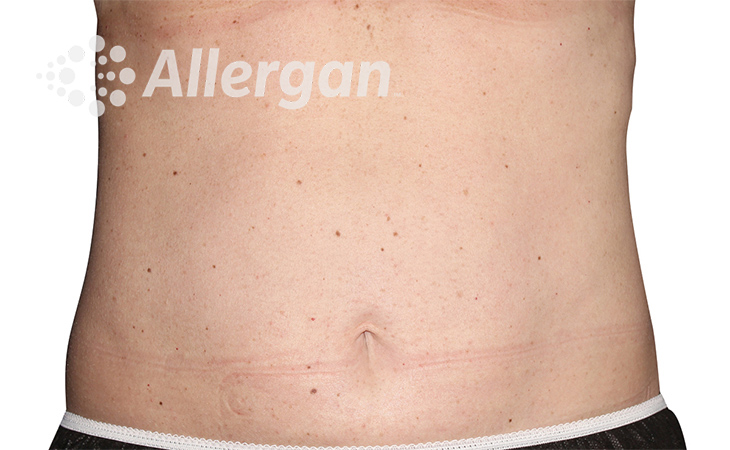
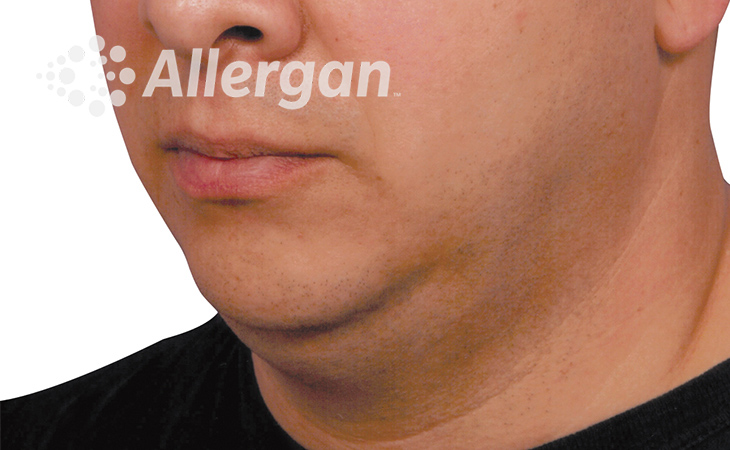
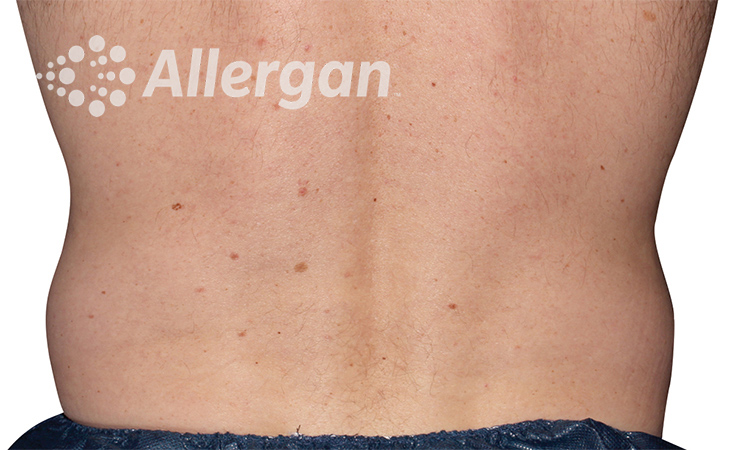
up to -27%(6)
US FDA-cleared14 for
NON-INVASIVE
BODY CONTOURING
EVERY Contour COUNTS


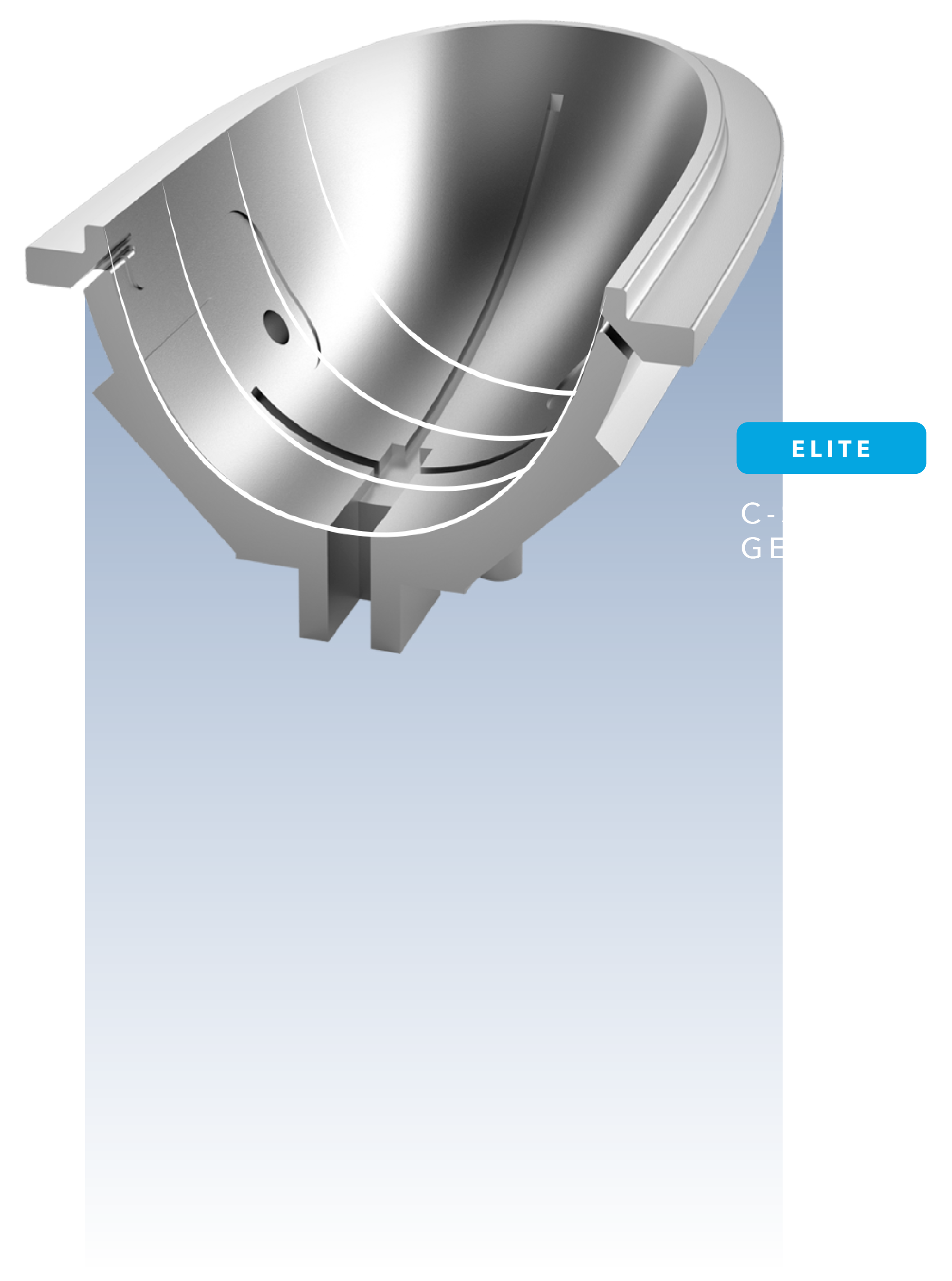
C
THE
DIFFERENCE
Better Fit for Better Results
Our new C-shaped cups are engineered to complement the body’s natural curves and to improve fit during tissue draw and features up to an 18% larger cooling area as compared to the previous generation.7,8
SAFETY FIRST
CoolSculpting® Elite is US FDA-approved14 and it has precise technologies in place to ensure optimal results under a safe procedure.



Freeze Detect®
Patented sensors are
designed to monitor tissue,
stopping treatment if a
potential freeze condition
is detected.9



Coolcontrol
Sensors track treatment
temperature in real time
and adjust the cooling
power to optimise
patient outcome.6



Gel Pad
As an additional measure, our thermal coupling gel pad provides consistent thermal contact between applicator and skin, to help protect your skin.9
HOW CoolSculpting® ELITE WORKS
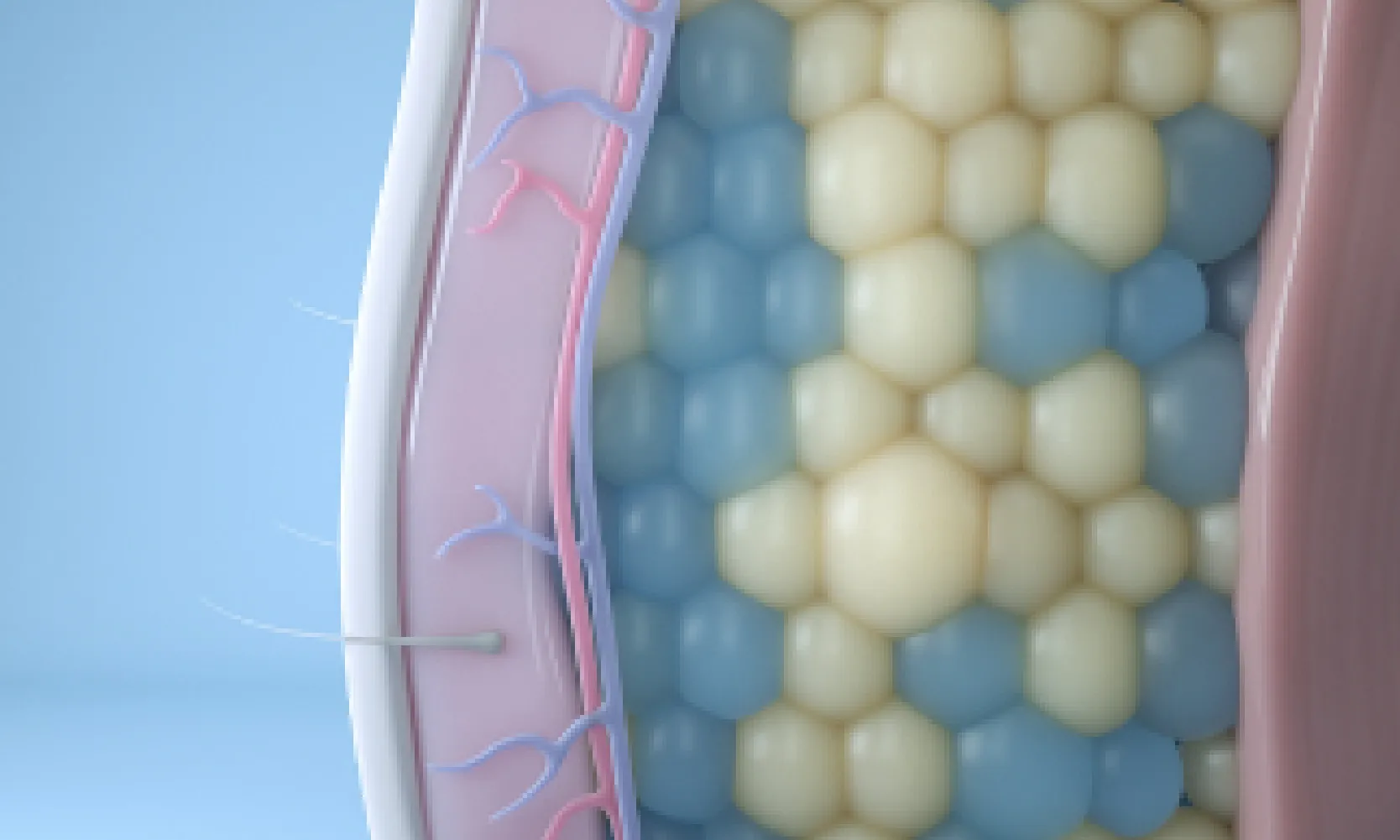


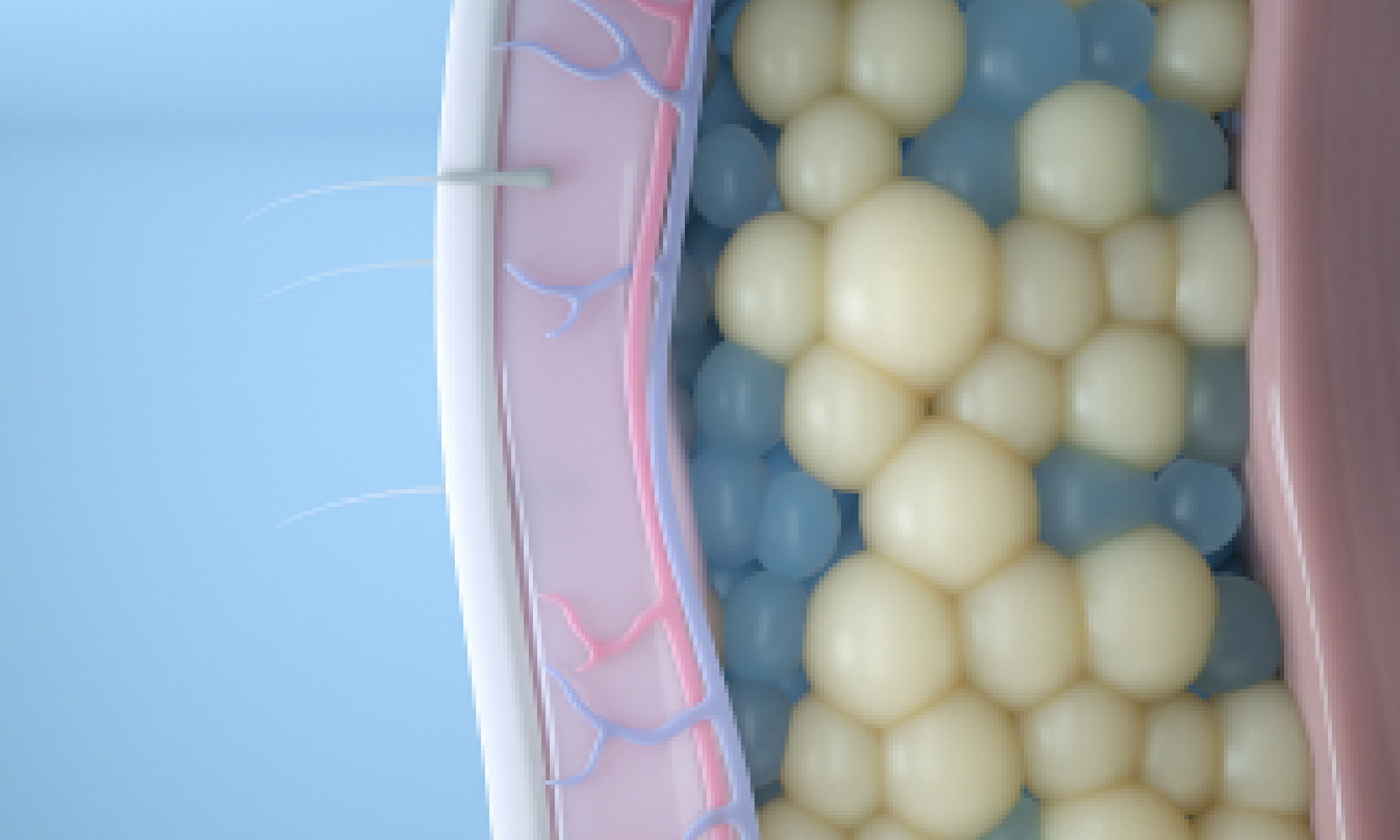
Target
We start by targeting and cooling fat cells to a temperature that will trigger apoptosis, or a natural cell death.4


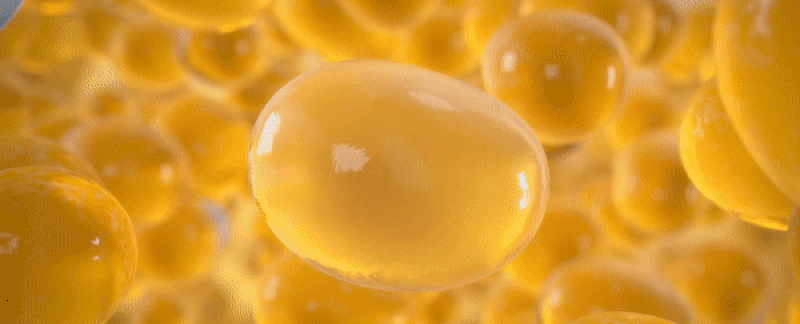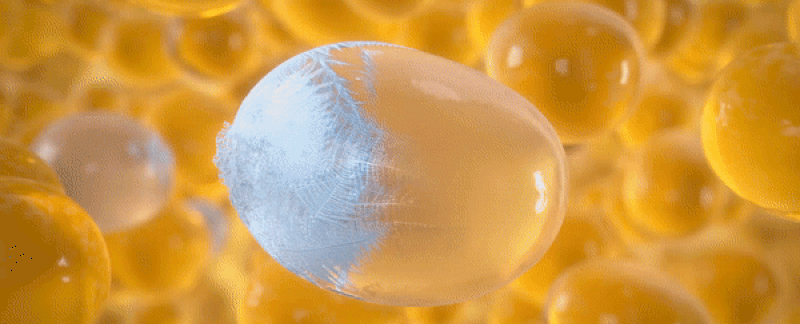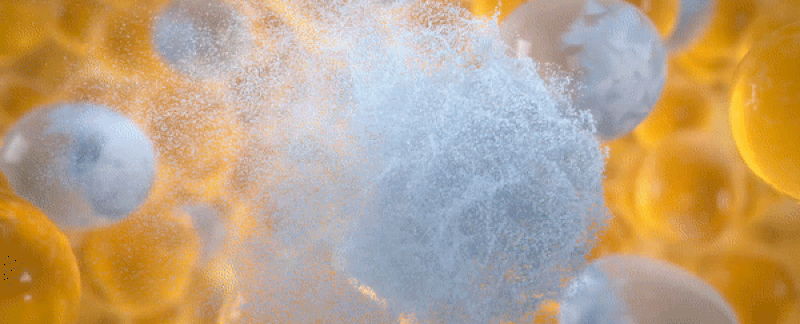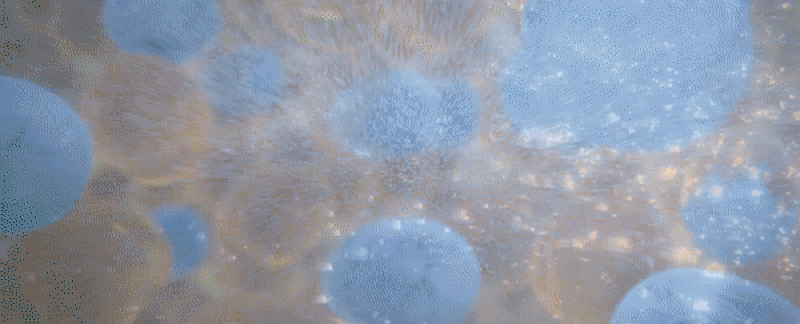
Freeze
Since fat is more sensitive to cold than the surrounding tissue, lipids in fat crystallize at a warmer temperature than water in other cell types.4
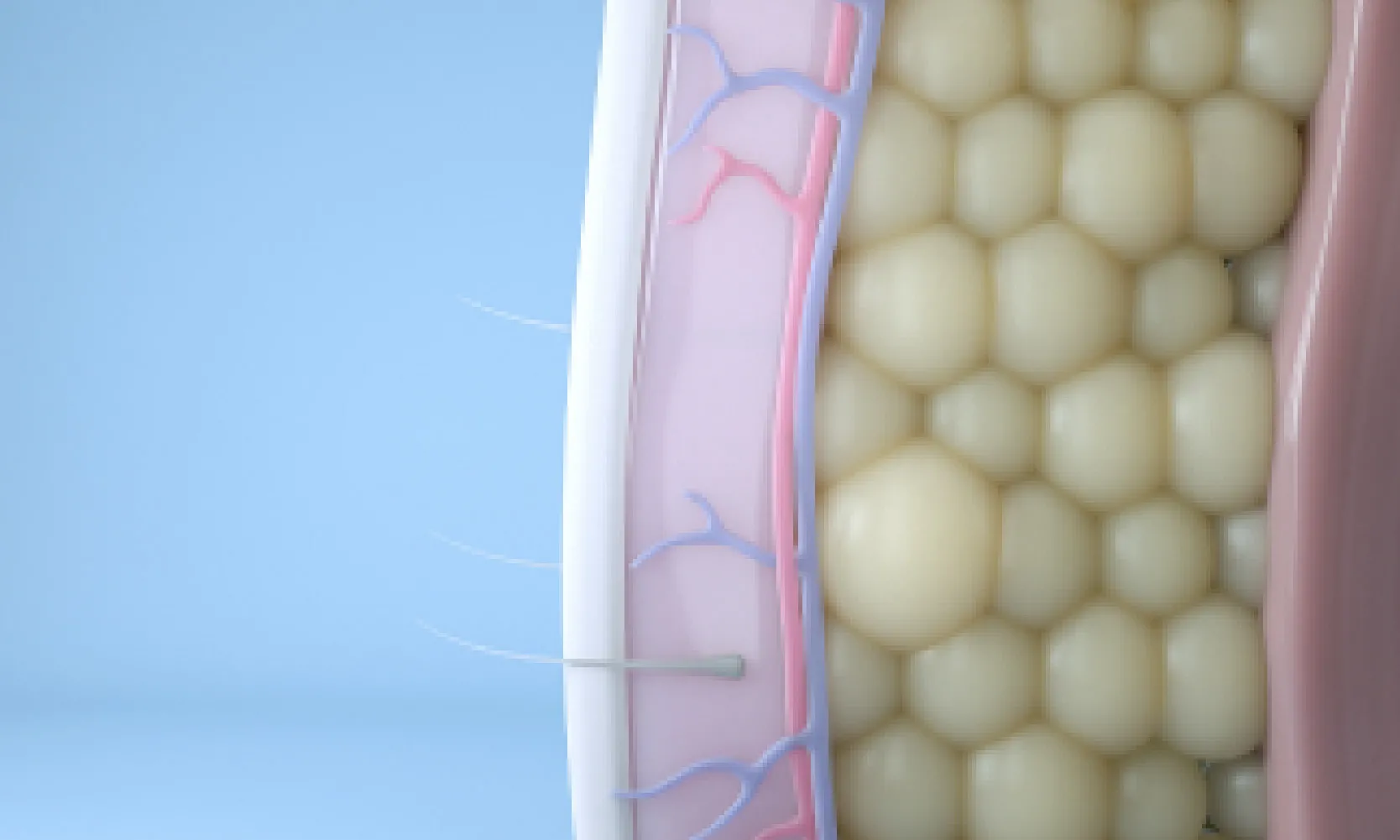


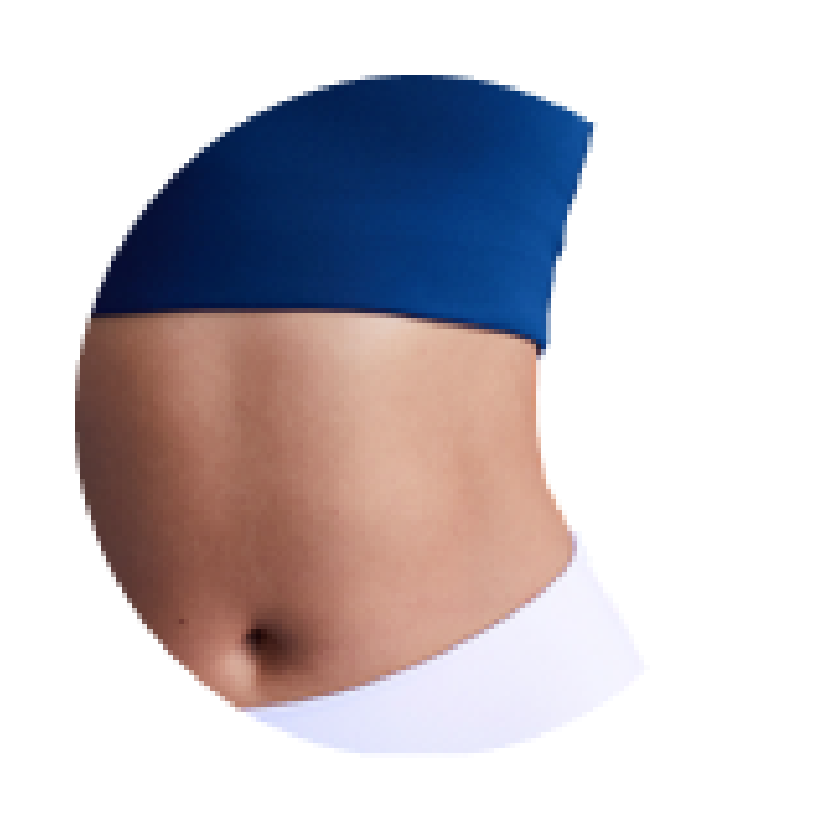
Reduce
Following treatment, body’s immune system will naturally eliminate dead cells which can result in up to 27% reduction of stubborn fat.6
 Back to the Top
Back to the Top
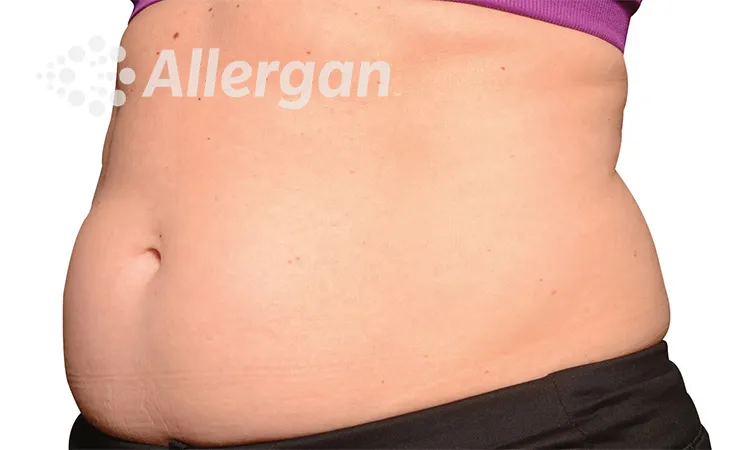
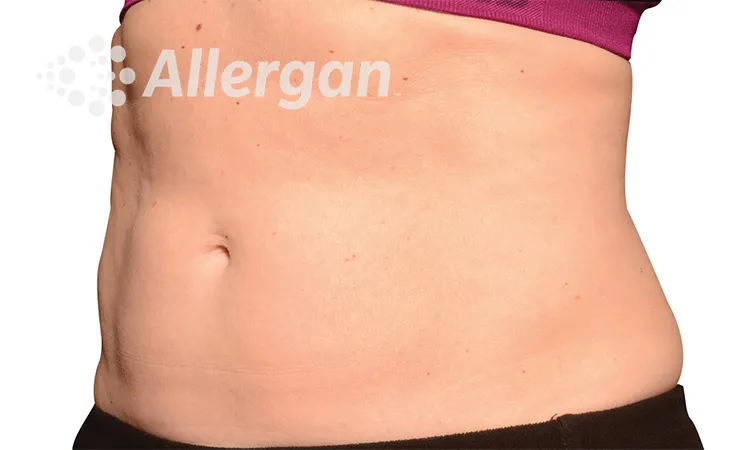
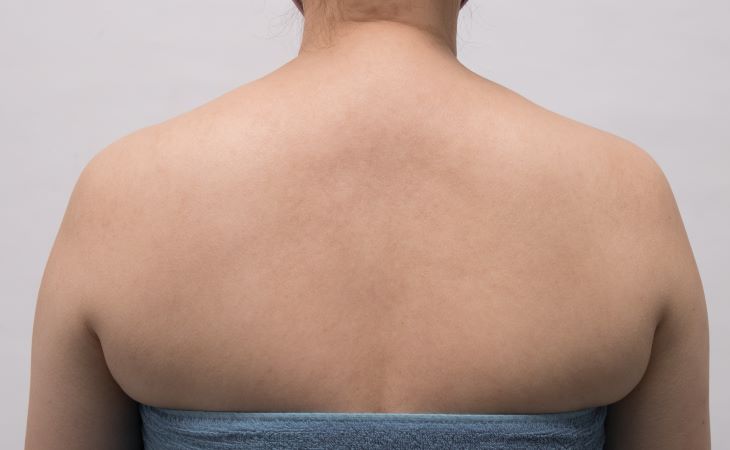
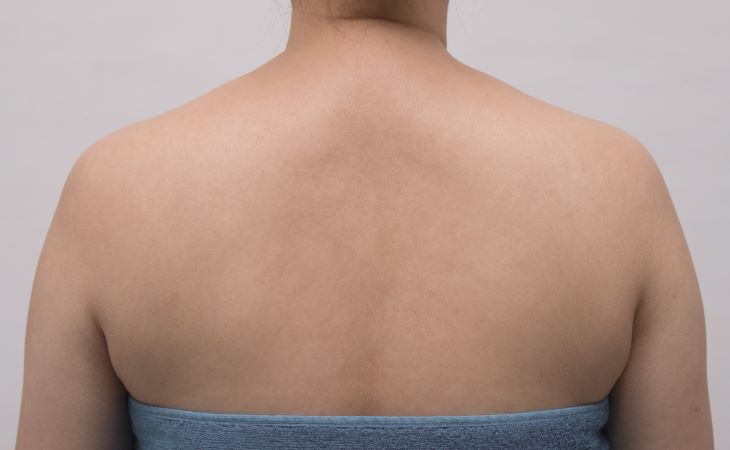
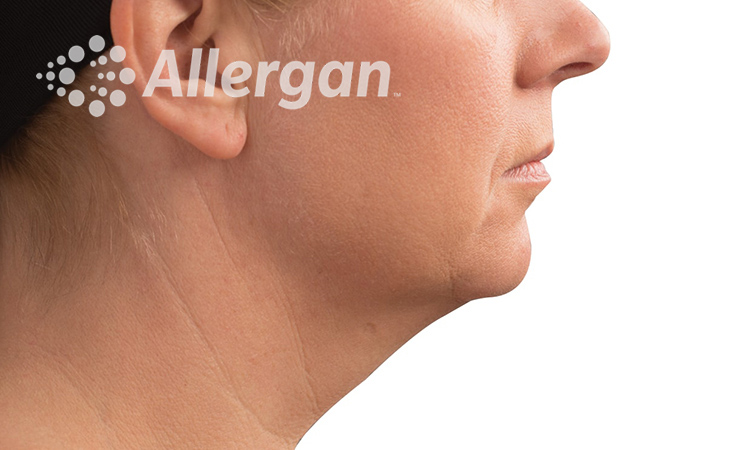
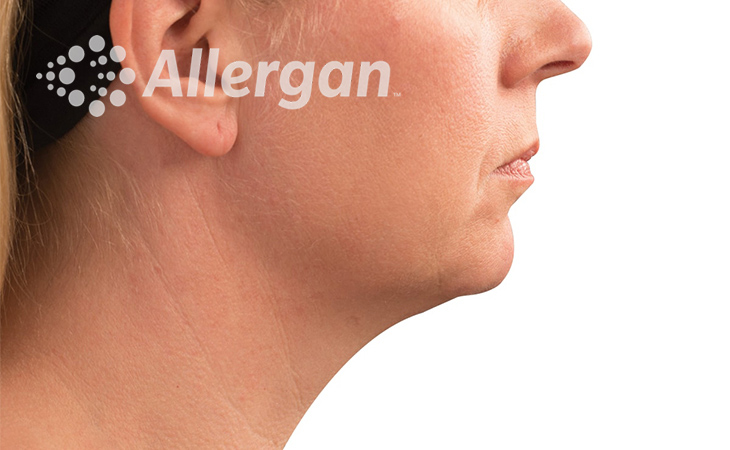
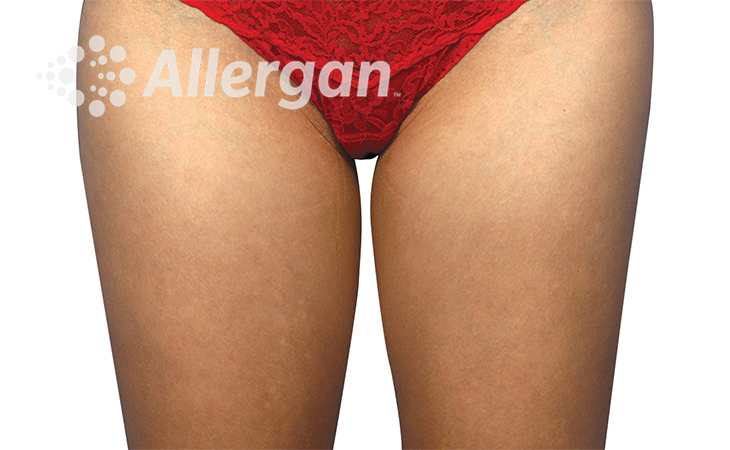
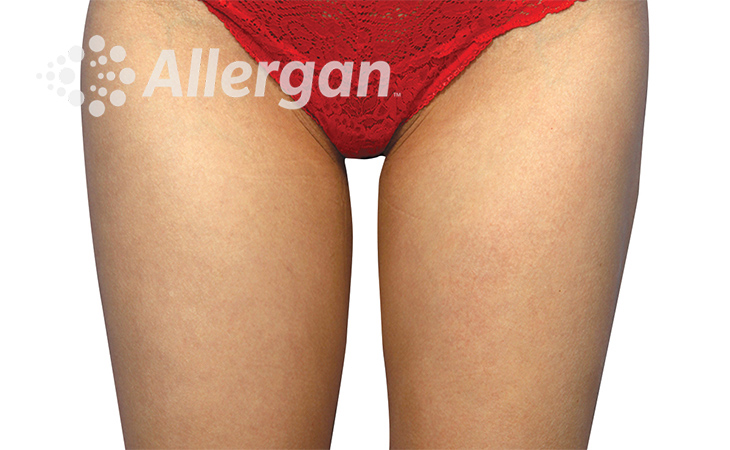
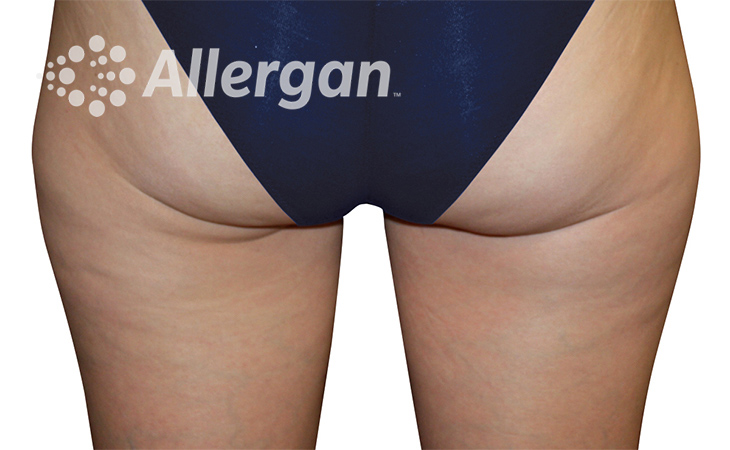
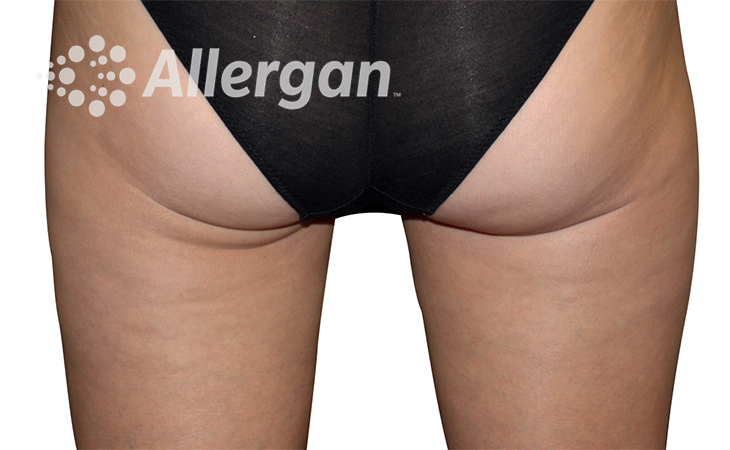
SEE REAL RESULTS
Click on a body area below to explore real CoolSculpting® transformations.
Ready to discover if CoolSculpting® Elite is right for you?
 Find a Clinic
Find a Clinic
Intended Use:
The CoolSculpting® Elite System is a non-invasive cooling and heating device that applies controlled cooling or heating to a treatment site on the patient's skin. Uses of the system in cooling mode include:
- Fat layer reduction through cold assisted lipolysis.
The optional massage function can also be used for temporary: - Reduction in the appearance of cellulite.
Important safety information:
During the procedure patients may experience sensations of pulling, tugging, mild pinching, intense cold, tingling, stinging, aching and cramping at the treatment site.13 These sensations subside as the area becomes numb.13
Following the procedure, typical side effects include redness, swelling, blanching, bruising, firmness, tingling, stinging, tenderness, cramping, aching, itching, skin sensitivity and numbness. Numbness can persist for up to several weeks. A sensation of fullness in the back of the throat may occur after submental treatment. The following rare and very rare side effects have the following incidence rates (approximate occurrences per number of treatments): paradoxical hyperplasia (1/3000 [0.033%]); late-onset pain (1/6000 [0.017%]); severe pain (1/6000 [0.017%]); hyperpigmentation (1/11000 [0.009%]); freeze burn (1/15000 [0.006%]); treatment area demarcation (1/20000 [0.005%]); vasovagal symptoms (1/30000 [0.003%]); subcutaneous induration (1/30000 [0.003%]); cold panniculitis (1/60000 [0.002%]) and hernia (1/185000 [0.001%]).13,15 The CoolSculpting® procedure is not for everyone. Patients should not have the CoolSculpting® procedure if they suffer from cryoglobulinaemia, cold agglutinin disease or paroxysmal cold haemoglobinuria.13 The CoolSculpting® procedure is not a treatment for obesity.14 CoolSculpting® is not a weight loss procedure and should not replace a healthy diet and active lifestyle.
Please see CoolSculpting® and CoolSculpting Elite® Important Safety Information for additional information.
Effect of treatment varies with individuals
References:
- Medical Insight Inc. The Global Aesthetic Market Study: Version XVII. 2019
- All time International Cycles Summary_040621. Data sourced from Connect Cube database [provided by International Business Excellence]. May 2021.
- Manstein D et al. Lasers Surg Med 2008; 40: 595-604
- Zelickson B, et al. Derm Surg 2009; 35:1462-70
- Allergan Inc. Unpublished Data. CoolSculpting® Publications. INT-CSC-2050471. December 2020.
- Sasaki GH, et al. Aesthet Surg J 2014; 34:420–31
- Allergan. Unpublished data. INT-CSC-2050028. CoolSculpting® clinical “Fit and Function” study to test the V003 wells. February 2020.
- Allergan. Unpublished data. INT-CSC-2050288. Applicator cooling and distribution dye test. August 2020.EN-A.
- CoolSculpting® System (CoolSculpting® Elite) user manual. CS-UM-CM3-07-EN-E. Feb 2023.
- Manstein D et al. Lasers Surg Med 2008; 40: 595-604
- Avram MM and Harry RS. Lasers Surg Med 2009; 41:703-8
- Krueger N, et al. Clin Cosmet Investig Derm 2014; 7:201-25
- CoolSculpting® system user manual. BRZ-101-TUM-SGP-B COM1. Sep 2021
- FDA 501(K) Summary Document, Available at : http://www.accessdata.fda.gov/ [Accessed February 2022].
- Allergan, Unpublished date, INT-CSC-2050028, CoolSculpting, clinical fit and function study testing of V003 cups, February 2020.
- Spalding KL,et al. Nature 2008; 453:783-7.
Disclaimer: CoolSculpting is not a weight loss treatment.
SG-CSC-230058 Date of Approval: October 2023
 It's not weight loss.jpeg)